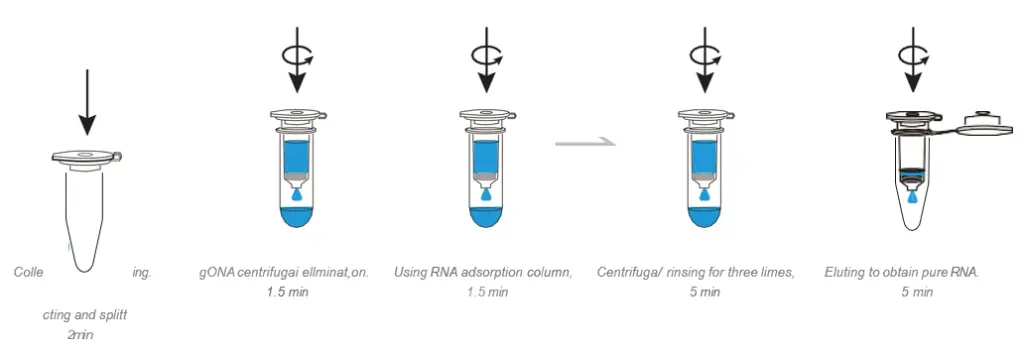
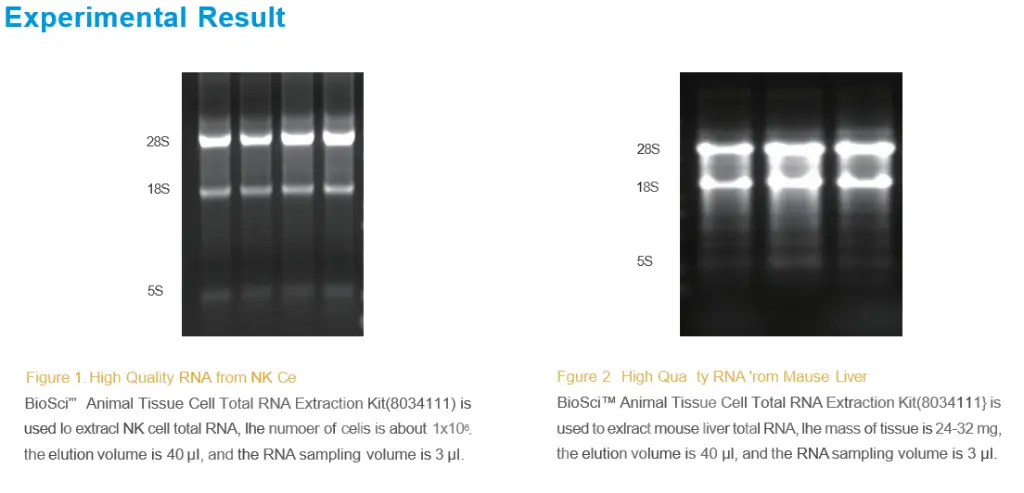
BioSci™ Animal Tissue Cell Total RNA Extraction Kit
Technical advantages :
1. No need of phenol and chloroform.
2. Unique lysate, no need of -mercaptoethanol.
3. Specific gDNA elimination column, effectively eliminating gDNA, without DNase digestion.
4. Repeated rinsing to ensure high purity. The A260/A280 ratio is 2,0-2.2.
5. Capable of extracting 10-100 ug of RNA.
6. Easy to use, the shortest time for test is 15-20 min.
Available at Gentaur
1. No need of phenol and chloroform.
2. Unique lysate, no need of -mercaptoethanol.
3. Specific gDNA elimination column, effectively eliminating gDNA, without DNase digestion.
4. Repeated rinsing to ensure high purity. The A260/A280 ratio is 2,0-2.2.
5. Capable of extracting 10-100 ug of RNA.
6. Easy to use, the shortest time for test is 15-20 min.
Available at Gentaur